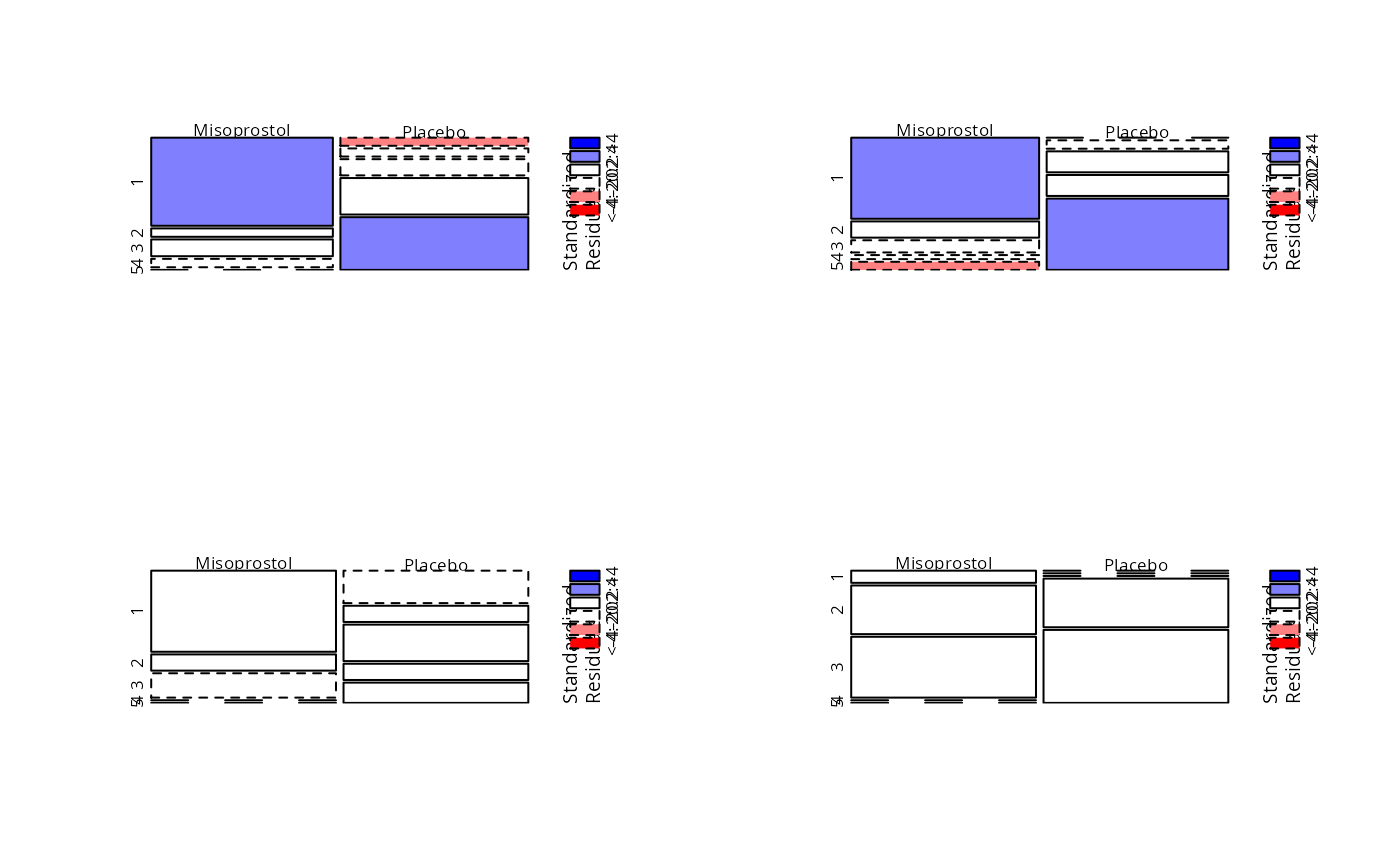
Prevention of Gastointestinal Damages
Lanza.RdData from four randomised clinical trials on the prevention of gastointestinal damages by Misoprostol reported by Lanza et al. (1987, 1988a,b, 1989).
Usage
data("Lanza")Format
A data frame with 198 observations on the following 3 variables.
studya factor with levels
I,II,III, andIVdescribing the study number.treatmenta factor with levels
MisoprostolPlaceboclassificationan ordered factor with levels
1<2<3<4<5describing an ordered response variable.
Source
F. L. Lanza (1987), A double-blind study of prophylactic effect of misoprostol on lesions of gastric and duodenal mucosa induced by oral administration of tolmetin in healthy subjects. British Journal of Clinical Practice, May suppl, 91–101.
F. L. Lanza, R. L. Aspinall, E. A. Swabb, R. E. Davis, M. F. Rack, A. Rubin (1988a), Double-blind, placebo-controlled endoscopic comparison of the mucosal protective effects of misoprostol versus cimetidine on tolmetin-induced mucosal injury to the stomach and duodenum. Gastroenterology, 95(2), 289–294.
F. L. Lanza, K. Peace, L. Gustitus, M. F. Rack, B. Dickson (1988b), A blinded endoscopic comparative study of misoprostol versus sucralfate and placebo in the prevention of aspirin-induced gastric and duodenal ulceration. American Journal of Gastroenterology, 83(2), 143–146.
F. L. Lanza, D. Fakouhi, A. Rubin, R. E. Davis, M. F. Rack, C. Nissen, S. Geis (1989), A double-blind placebo-controlled comparison of the efficacy and safety of 50, 100, and 200 micrograms of misoprostol QID in the prevention of ibuprofen-induced gastric and duodenal mucosal lesions and symptoms. American Journal of Gastroenterology, 84(6), 633–636.